Blackbody
A blackbody is an idealized physical entity in physics that absorbs all incident electromagnetic radiation and emits the maximum possible radiation for its temp...
Blackbody radiation is the thermal electromagnetic emission from an ideal object that absorbs and emits all energies, with a spectrum set solely by temperature.
Blackbody radiation is the electromagnetic radiation emitted by an idealized object, called a blackbody, which absorbs all incident electromagnetic energy and re-emits energy in a spectrum determined solely by its absolute temperature. A perfect blackbody reflects no light and transmits nothing; it is the ultimate absorber and emitter. The concept is foundational in thermodynamics, quantum mechanics, and astrophysics, providing a universal reference for understanding how real objects radiate energy.
At the atomic level, blackbody radiation results from the random thermal motions of charged particles, especially electrons. These motions cause emission of electromagnetic waves across a continuous range of wavelengths. Although no material is a perfect blackbody, many objects (such as lamp black, cavity radiators, or stars) closely approximate this ideal.
A blackbody is defined as a perfect absorber—it takes in all electromagnetic radiation, regardless of wavelength or angle, with no reflection or transmission. Equally, it is a perfect emitter, radiating the maximum possible energy at each wavelength for a given temperature. In thermal equilibrium, a blackbody’s absorbed and emitted energies are balanced, so its temperature remains constant.
Real-world approximations are achieved with cavities having small holes: radiation entering the cavity is absorbed after many internal reflections, mimicking a blackbody. This principle is used in laboratory blackbody sources.
Thermal radiation is electromagnetic radiation generated from the thermal motion of particles in matter. For a blackbody, this is the maximum emission possible for its temperature. As temperature increases, total emitted energy and the frequency of peak emission both rise.
Thermal radiation is the dominant energy transfer mode in vacuums (like space), and it’s crucial for remote sensing, temperature measurements, and energy balance in physics and engineering.
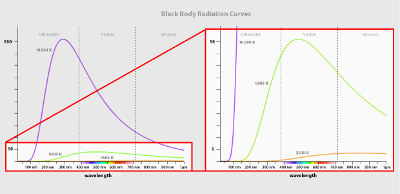
Blackbody radiation forms a continuous spectrum with energy at every wavelength, unlike the discrete lines seen in atomic emission/absorption. The distribution of energy across wavelengths is smooth, with a characteristic curve for each temperature.
The spectral shape is dictated by Planck’s law. As temperature rises, the emission peak shifts to shorter wavelengths (Wien’s law), and the total emitted energy increases sharply (Stefan-Boltzmann law).
Emissivity (( \epsilon )) measures how closely a real surface matches a perfect blackbody in emitting thermal radiation. Defined as:
[ \epsilon = \frac{\text{Emitted intensity by real surface}}{\text{Emitted intensity by blackbody}} ]
A perfect blackbody has ( \epsilon = 1 ); real objects have ( \epsilon < 1 ), with values depending on material, surface finish, and wavelength. Knowledge of emissivity is essential for accurate infrared temperature measurements and thermal engineering.
The study of blackbody radiation drove pivotal advances in physics:
These breakthroughs revolutionized understanding of energy, matter, and electromagnetic waves.
Planck’s law gives the spectral radiance of a blackbody at temperature ( T ):
[ B_\lambda(T) = \frac{2\pi hc^2}{\lambda^5} \cdot \frac{1}{e^{hc/(\lambda k_B T)} - 1} ]
where:
This law matches experimental results at all wavelengths and revealed the quantum nature of energy.
The peak wavelength ( \lambda_{\text{max}} ) of blackbody emission is inversely proportional to temperature:
[ \lambda_{\text{max}} T = b ]
where ( b = 2.897771955 \times 10^{-3} ) m·K. Hotter objects emit peak radiation at shorter (bluer) wavelengths.
The total energy radiated per unit area is:
[ j^* = \sigma T^4 ]
where ( \sigma = 5.670374419 \times 10^{-8} ) W·m⁻²·K⁻⁴. This shows energy emitted rises rapidly with temperature.
The classical Rayleigh-Jeans law predicts:
[ B_\lambda(T) = \frac{2 c k_B T}{\lambda^4} ]
At short wavelengths, this formula diverges (predicts infinite energy), which is not observed—this is the ultraviolet catastrophe. Planck’s law fixed this by introducing quantized energy.
Blackbody spectra are plotted as intensity versus wavelength for various temperatures:
Key features:
For example, the Sun’s surface (~5778 K) peaks in the visible range; at room temperature (~300 K), emission peaks in the infrared.
Stars behave as near-perfect blackbodies. Their color reveals surface temperature: blue stars are hottest, red stars are cooler. By fitting observed spectra to blackbody curves, astronomers estimate stellar temperatures and sizes.
A cavity with a tiny aperture acts as a practical blackbody. Light entering the hole is absorbed after many reflections, and the emission from the hole matches the theoretical blackbody spectrum. Such devices calibrate scientific instruments.
The CMB is the afterglow of the Big Bang, with a near-perfect blackbody spectrum at 2.725 K. Measuring its spectrum has validated cosmological models and the Big Bang theory.
Applications include:
Blackbody models estimate stellar and planetary properties, energy outputs, and aid in classifying stars. Emission from galactic dust and planetary atmospheres is analyzed using blackbody curves.
Instruments infer temperatures from infrared emissions compared to blackbody standards. Used in industry (furnaces, engines), medicine (fever screening), and earth observation (climate satellites).
Earth is modeled as an imperfect blackbody for energy balance studies. The greenhouse effect arises from atmospheric gases altering the planet’s emissivity and radiative properties.
Non-contact pyrometry, thermal insulation, radiative cooling systems, and solar absorber design all rely on blackbody theory for optimization and safety.
The study of blackbody radiation was crucial in the development of quantum mechanics. Classical physics could not explain the actual spectrum (ultraviolet catastrophe), but Planck’s quantization of energy provided the correct formula, revolutionizing our understanding of light, matter, and energy.
Blackbody radiation remains a touchstone in physics, engineering, and astronomy—helping to calibrate instruments, infer temperatures, and set the stage for quantum theory.
References:
For further reading, see fundamental physics textbooks and specialized literature on thermodynamics and quantum mechanics.
A blackbody is a theoretical object that perfectly absorbs all electromagnetic radiation incident upon it, regardless of wavelength or angle. It also re-emits radiation at the maximum possible intensity for a given temperature, with a spectrum determined solely by its temperature.
Blackbody radiation is foundational in physics as it led to the development of quantum mechanics. The inability of classical physics to explain its spectrum (the ultraviolet catastrophe) prompted Planck's quantum hypothesis. Blackbody models are also essential in astrophysics, thermal engineering, and climate science.
Real objects have an emissivity less than one, meaning they do not emit as much radiation as a perfect blackbody at the same temperature. Their emission can also depend on wavelength, surface roughness, and material properties, unlike the ideal blackbody.
By measuring the spectrum of light a star emits, astronomers identify the peak wavelength of emission. Using Wien's displacement law, the star's surface temperature can be calculated based on this peak.
The ultraviolet catastrophe refers to the prediction by classical physics (Rayleigh-Jeans law) that a blackbody would emit infinite energy at short wavelengths (ultraviolet), which is not observed. Planck resolved this by introducing quantized energy levels, giving rise to quantum mechanics.
Understand how blackbody radiation shapes quantum theory, astrophysics, and modern technology. Learn how this fundamental concept influences everything from star colors to thermal imaging.
A blackbody is an idealized physical entity in physics that absorbs all incident electromagnetic radiation and emits the maximum possible radiation for its temp...
Thermal radiation refers to electromagnetic radiation emitted by matter due to its temperature, occurring even in a vacuum. It underlies phenomena like the warm...
Radiant energy is the energy carried by electromagnetic radiation, spanning the electromagnetic spectrum from radio waves to gamma rays. It is crucial in fields...
Cookie Consent
We use cookies to enhance your browsing experience and analyze our traffic. See our privacy policy.