Temperature
Temperature is a fundamental physical quantity representing the average kinetic energy of the particles in a substance. Measured in kelvins (K), it underpins th...
Kelvin (K) is the SI unit of thermodynamic temperature, defined by the Boltzmann constant, and essential for universal, absolute temperature measurement in science and engineering.
The kelvin (symbol: K) is the fundamental SI unit of thermodynamic temperature, underpinning all scientific and engineering temperature measurements. Defined since 2019 by fixing the Boltzmann constant (k) at exactly 1.380 649 × 10⁻²³ J·K⁻¹, the kelvin provides a direct link between temperature and microscopic energy. This definition, rooted in immutable physical constants, ensures that the kelvin is universally stable, reproducible, and independent of any material substance or artifact.
Unlike Celsius and Fahrenheit, the kelvin is an absolute scale: it begins at absolute zero (0 K), the theoretical point where a system’s entropy and thermal motion reach their minimum. The magnitude of one kelvin interval is identical to one degree Celsius, but the scales differ in their starting points.
The kelvin’s role as an SI base unit makes it central to physical law, thermodynamics, metrology, engineering, and especially aviation and aerospace applications, where precise, traceable temperature knowledge is vital for safety and performance.
The kelvin is defined as:
The kelvin, symbol K, is the SI unit of thermodynamic temperature. It is defined by taking the fixed numerical value of the Boltzmann constant k to be 1.380 649 × 10⁻²³ joule per kelvin (J·K⁻¹).
Why is this important?
By tying the definition to the Boltzmann constant and, via the joule, to the SI units of mass (kg), length (m), and time (s), the kelvin is anchored in the very fabric of physical law. This ensures temperature is universally measurable, accurate, and stable, regardless of time or location.
| Constant | Symbol | Value | Unit |
|---|---|---|---|
| Boltzmann constant | k | 1.380 649 × 10⁻²³ | J·K⁻¹ |
| Planck constant | h | 6.626 070 15 × 10⁻³⁴ | J·s |
| Hyperfine transition (Cs) | Δν_Cs | 9 192 631 770 | Hz |
The Kelvin scale starts at absolute zero (0 K; −273.15°C), where all classical molecular motion ceases. Every unit interval (1 K) is the same as one degree Celsius. The Kelvin scale is linear and unbounded above, making it essential for scientific equations, quantum mechanics, and thermodynamics.
Key points:
Absolute zero (0 K) is the theoretical temperature where a system’s entropy is lowest and classical motion ceases. It is unreachable in practice, but modern cryogenic and laser cooling techniques can approach it within billionths of a kelvin.
Why does it matter?
The Boltzmann constant (k) bridges the microscopic world (energy per particle) and the macroscopic world (temperature). It is foundational in statistical mechanics and thermodynamics.
Fixing k’s value in the SI definition of kelvin ensures that temperature is based on fundamental physics, not material artifacts.
Thermodynamic temperature is the absolute, substance-independent measure of temperature. It determines the direction of heat flow and spontaneous processes, and is used in all core thermodynamic equations (e.g., Carnot efficiency, entropy, Gibbs free energy).
The kelvin is one of the seven fundamental SI base units. Its definition, via the Boltzmann constant, relates it directly to the kilogram, metre, and second:
[ 1,\text{J} = 1,\text{kg} \cdot \text{m}^2 \cdot \text{s}^{-2} ]
This ensures universal traceability and compatibility with all SI-based measurements.
Derived SI quantities using kelvin:
Kelvin must be used in scientific formulas where temperature is an absolute measure:
Milestones:
This constant-based definition enhances universality and eliminates reliance on material reference points.
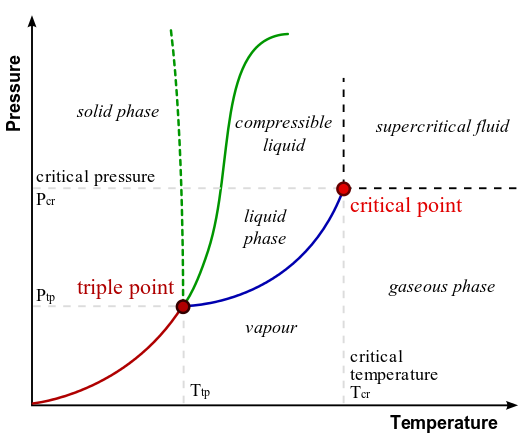
The triple point of water (273.16 K, 611.657 Pa) is where water’s solid, liquid, and vapor phases coexist. It was the historical reference for kelvin’s definition. Now, it remains vital for calibration, especially using Vienna Standard Mean Ocean Water (VSMOW) for isotopic consistency.
VSMOW defines the precise isotopic composition of water used as a reference in temperature calibration, ensuring reproducibility and consistency worldwide.
Conversions:
| Event | Kelvin (K) | Celsius (°C) | Fahrenheit (°F) |
|---|---|---|---|
| Absolute zero | 0 | −273.15 | −459.67 |
| Triple point (water) | 273.16 | 0.01 | 32.02 |
| Freezing (water) | 273.15 | 0 | 32 |
| Boiling (water) | 373.15 | 100 | 212 |
After the 2019 redefinition, kelvin is realized by:
National metrology institutes disseminate temperature standards and calibrations in kelvin.
A phase diagram shows the regions where water is solid, liquid, or vapor, and highlights the triple point (273.16 K), freezing point (273.15 K), and boiling point (373.15 K). It is essential for calibration and understanding environmental control in aviation and science.
Kelvin (K) is the cornerstone of temperature measurement in science, engineering, and technology. Its definition, based on the Boltzmann constant, ensures that it is universal, stable, and tied to the fabric of physical law. As an SI base unit, it is indispensable for accurate, reproducible, and meaningful temperature data—essential for modern applications from aviation to quantum research.
Kelvin is absolute because it starts at absolute zero—the point where all thermal motion ceases, according to the laws of physics. This makes it independent of arbitrary reference points, unlike Celsius or Fahrenheit, and essential for thermodynamic calculations.
Kelvin is realized using experiments based on fundamental physical constants, such as acoustic gas thermometry, Johnson noise thermometry, and dielectric constant gas thermometry. These methods link temperature to the Boltzmann constant, enabling universal and reproducible measurements.
Kelvin and Celsius scales have identical intervals; 0 K is −273.15°C. The formulas are: T(°C) = T(K) − 273.15 and T(K) = T(°C) + 273.15. Fahrenheit relates via T(°F) = (9/5)T(°C) + 32 and T(°F) = (9/5)(T(K) − 273.15) + 32.
The Boltzmann constant (k) connects microscopic energy (per particle) to macroscopic temperature. By fixing its value, the kelvin is linked directly to fundamental physics, ensuring stability and reproducibility without relying on material artifacts.
Kelvin is vital for calibrating temperature sensors, managing environmental control systems, performing engine thermodynamics, and ensuring safety in extreme conditions, such as high altitudes or space, where absolute temperature knowledge is crucial.
Kelvin is fundamental for aviation, engineering, and scientific research. Let us help you implement SI-traceable temperature solutions for your applications.
Temperature is a fundamental physical quantity representing the average kinetic energy of the particles in a substance. Measured in kelvins (K), it underpins th...
Ambient temperature, crucial in aviation and meteorology, is the temperature of undisturbed air surrounding an object, measured per ICAO and WMO standards. It f...
A kilowatt (kW) is a standard unit of power equal to 1,000 watts, used globally to measure electrical power in systems from household appliances to aviation gro...
Cookie Consent
We use cookies to enhance your browsing experience and analyze our traffic. See our privacy policy.